DisFact #12: Dodgy medical devices; Macroeconomic tracker; Indian Facebook users
Welcome to DisFact. I am Samarth Bansal. Thanks for subscribing. Know a friend who would enjoy reading this newsletter? Please consider forwarding this email. Here is the sign up link and list of all previous issues.
Correction: In last week’s issue, I had compared ad spending figures for NDA (2014-now) and UPA(2004-14) government. Two readers wrote back and rightly pointed out that the comparison was inappropriate as the spending amount had not been adjusted for inflation. I apologise for the oversight. I will write about this again when I discuss campaign finance (soon).
#ImplantFiles: the murky world of medical devices
In the second issue of DisFact, I wrote about the scandalous case of Johnson & Johnson faulty hip implants:
In August 2010, Johnson & Johnson’s (J&J) faulty hip implants were recalled globally as “it left large quantities of metal debris in the body and it had a very high failure rate”. 4,500 patients in India were implanted with the faulty hip joint. But neither the company nor the regulators systematically reached out to patients to inform them of the faulty implant. The victims continued to suffer.
This week, a ten-month long investigation by the Indian Express—carried out with more than 250 reporters and data specialists from 36 countries under the International Consortium of Investigative Journalists—revealed that J&J’s was not a one-off case: it’s a systemic problem that plagues the global health care industry, thanks to the dubious nexus between companies, doctors and hospitals, highlighting significant flaws in our health regulatory system. It’s frightening.
What we learnt from #ImplantFiles: “Almost every medical device, from coronary stents and pacemakers to breast and knee implants, is advertised, sold, and surgically implanted with barely any oversight. The medical device industry is booming but transparency is lacking.”
The global cost of dodgy devices:
Medical devices that broke, misfired, corroded, ruptured or otherwise malfunctioned after implantation or use — despite assurances by regulators, the industry and doctors that they were safe — were linked to 1.7 million injuries and nearly 83,000 deaths over the last decade. Nearly 5,00,000 reports mentioned an explant — a surgery to remove a device — in connection with an adverse event.
Read: Everything You Need to Know About the Implant Files (ICIJ)
In India, almost 70% of medical devices are imported—but without any checks. The medical device industry is currently valued at $5.2 billion and contributes 4-5% to the $96.7 billion Indian health care industry.
Issues:
1. Rise in the number of reported incidents: The number of “medical device adverse events”, such as deaths after the installation of a stent, has gone up from 40 in 2014 to 556 so far this year. This is an underestimate. (Read more)
2. Low reporting of device failure: “Though the reporting of device failure began in 2014, the results are still not being made public. Complaints of a very serious nature are fed into a lone computer in an obscure office in Ghaziabad.”
Basically, the Central Drugs Standard Control Organisation (CDSCO), the regulator, does not maintain a public list of devices that have been recalled from the global or Indian markets.
Why this information needs to be public: so that patients can make informed decisions.
3. Around half of the devices recalled in other countries were being sold in India: As many as 117 devices have been recalled over the past two years by the United States Food and Drug Administration after it concluded that the devices led to serious health issues, including death. However, at the time of recall, at least 57 of these devices were still being marketed in India to patients. (Read more)
The Indian Express tracked eight of these devices that are the most commonly used, and found that in each case, it was left to the manufacturer to take action. In some cases, the manufacturers claimed that the recall was due to technical defects while in others, they said the software was to blame. All eight devices are back in the market.
More specifics here: In US, ortho major forced to admit it was corrupting doctors and hospitals in India
4. What should medical device manufacturers do when problems are found: It is their responsibility to track down the recipients of withdrawn medical devices, tell them about the problems, and pay compensation. But companies are not doing so.
This example shows the state of regulation in India: the regulator didn’t even know about a case where devices were recalled.
Many users of Medtronic’s insulin pump were notified in September 2017 about the device being recalled internationally. The users of the insulin pumps - small electronic devices that infuse insulin into a body through the day - said company representatives met with them and replaced their devices. However, regulator Central Drugs Standard Control Organisation (CDSCO), has no record of this recall ever having taken place. The CDSCO is meant to regulate recalls by displaying “medical advice alerts” so that patients can know when devices are being recalled.
Medtronic recalled the devices in 2017 saying they could trigger an excessive amount of insulin in a user’s body, resulting in possible hypoglycaemia (better known as low blood sugar). Around 10,000 devices were recalled. However, senior diabetologists in Delhi say Medtronic wasn’t proactive in recalling the devices. They said they were the ones who noticed the news of the recall by the US Food and Drug Administration and informed Medtronic in India about it.
5. How companies pull it off: Medtronic, the world’s largest cardiac devices company, may have indulged in a range of irregularities to push their products in India. “Doctors were given freebies, unsuspecting patients were given these products, loans were granted to fund surgeries, and crores were spent on seminars to push products.” (Read more)
6. What are the redress mechanisms if devices found faulty?
Currently, there are no specific rules or a law that allows the government to direct a medical device company to give compensation to a victim after the device has been found faulty. Following the emergence of faulty hip implants from Johnson & Johnson, the Ministry of Health and Family Welfare is working on a formula for compensation. There are proposals that the government might include a special clause under the Medical Device Rules to include compensation for patients. But the patient can always approach a consumer court to seek damages from the device company.
A Business Standard editorial raises an important question:
The deeper question, however, is whether such misuse is an inevitable consequence of a tertiary health care system that relies excessively on the private sector. A large and vibrant public sector in tertiary health care is the only way to counteract the hurtful consequences of information asymmetries and poor regulation, especially in sectors such as medical devices, pathological tests, and so on. The government should re-examine its plan for universal health care that chooses to give too much space to a health care sector that has shown itself to be insufficiently trustworthy.
Read more: All #ImplantFiles stories from the Indian Express (Click here)
How is the economy really doing?
In October, Mint’s data team started tracking 16 high-frequency indicators to assess the health of the Indian economy in a monthly macroeconomic tracker.
Why track high-frequency indicators?
As India’s GDP numbers are released with a lag of three months and given the persistent doubts surrounding the calculations of the new GDP series, it becomes important to track other high-frequency indicators to gauge the state of the economy.
What will it capture:
The 16 indicators capture trends across four aspects of the economy: consumer spending, industrial activity, external vulnerability and “ease of living” (which measures inflation and the jobs scenario).
Mint compares the performance of recent months with the long-term five-year trend.
What the latest data says: Nine of the 16 indicators are in the red—performing worse than the five-year trend. Indian economy remains weak despite oil relief. (Read more here)
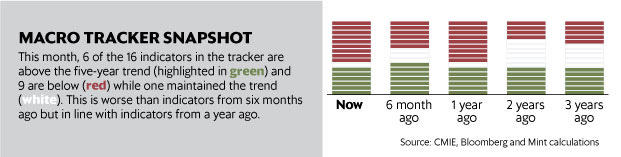
Key points:
“The biggest concerns are in the external sector where all four indicators [import cover, currency, current account balance, trade balance] continue to perform below the five-year trends even with recent positive developments.”
“Another potential area of concern is consumer activity. Growth in passenger vehicle sales, tractor sales and domestic air passengers are all below five-year trends. And while, two-wheeler sales outperformed trend growth, this is partly a reflection of underperformance in the base year.”
“One area where the economy is doing better is industry. Industrial production in the economy continued to grow at a healthy pace during the July-September quarter and in the subsequent month.”
Mint releases the findings from its tracker at the end of every month. Going forward, I will share the key takeaways in this newsletter.
Whom can Indian political parties reach on Facebook and why it matters
My story this week: Arjun Srinivas and I used data from the Facebook advertising platform to find out demographic breakup of Indian FB users and what it means for electoral politics.
Facebook is used by 270 million Indians in the age group of 18-65 monthly, meaning political parties can reach an estimated 36% of the voting age population on the platform. And the user base is growing: the number of Facebook users in India have more than doubled since the previous general election in 2014, according to data portal Statista.
Indian Facebook users, however, are skewed towards specific demographics. A Hindustan Times analysis of anonymous and aggregated data from Facebook’s advertising portal shows that the bulk of the platform’s Indian users are men, under the age of 30 and live in urban clusters.
Key points:
77% of the Indian Facebook profiles belong to men, compared to 57% globally.
Young people dominate the platform: 63% of all users are below the age of 30.
Facebook demographics vary across states. Close to 63% of the total population (age group 20-65) in Delhi-National Capital Region (NCR) is estimated to be on Facebook, while only 17.8% of Bihar is.
Around 81% of the current Indian Facebook users accessed the network through a high-speed 4G connection.
Talk to me
Comments? Feedback? Suggestions? Write to me at samarthbansal42@gmail.com or hit reply to this email. Thank you!

